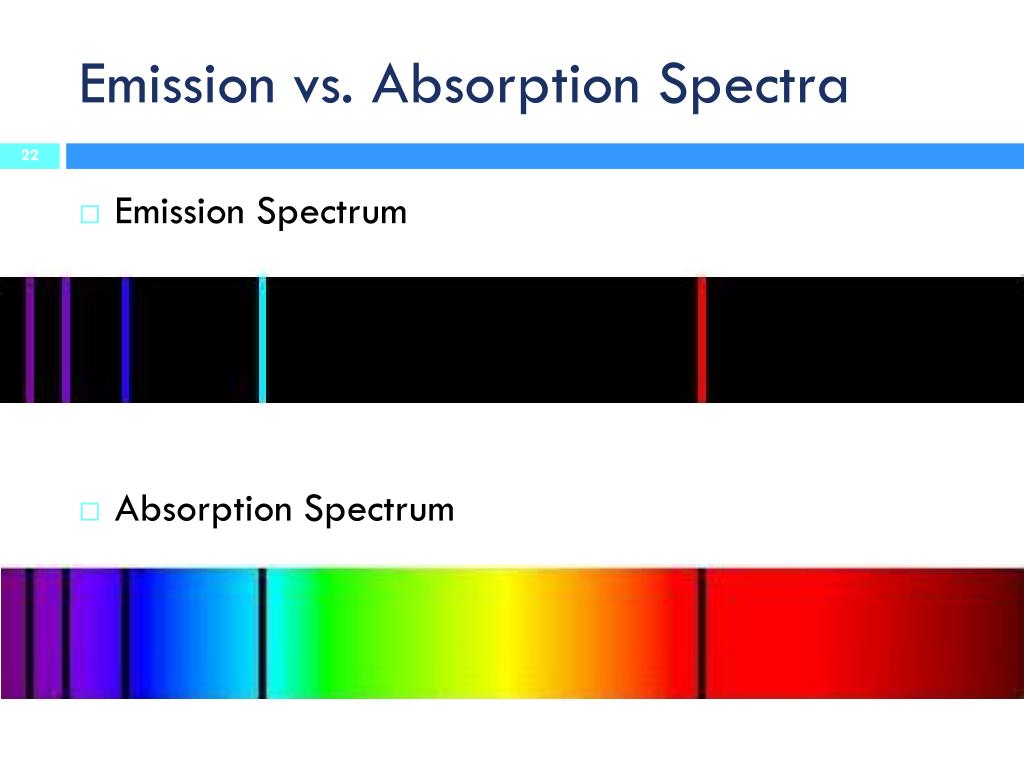
That term makes the equation impossible to solve analytically.įor hydrogen, the energy of an energy level is determined solely by its principal quantum number. The term for the electron-electron repulsion makes things difficult because both electrons can be moved. However, for higher atoms such as helium, where there are more than one electron, the Schrödinger equation contains two terms for the two nucleus-electron attractions and a term for the electron-electron repulsion. Also, the nucleus is not moving, which simplifies some calculations We only need to consider the interaction between those two particles. This is possible because we only need to consider two particles: the nucleus which has a positive charge and the electron which has a negative charge. $E_n$ is the energy of the $n$-th energy level.The dark bands in the absorption spectrum and the colored bands in the emitted spectrum of the same element are similar.We have solved the Schrödinger equation for hydrogen-like atoms such as $\ce$. But in an absorption spectrum there’ll be few dark bands within the continuous spectrum. In an emission spectrum there’ll be only few colored bands in a dark back ground.Compared to the continuous visible spectrum, both emission and absorption spectra are line spectra because they only contain certain wavelengths.In simple terms, absorption spectra records the wavelengths absorbed by the material, whereas emission spectra records wavelengths emitted by materials, which have been stimulated by energy before.This type of spectrum is called an emission spectrum. When the species come back to the ground state from the excited state, the absorbed radiation is emitted, and it is recorded.When an atom or molecule excites, it absorbs a certain energy in the electromagnetic radiation therefore, that wavelength will be absent in the recorded absorption spectrum. 
What is the difference between Absorption spectra Vs emission spectra? So the absorption spectrum is quite complex, and the molecule absorbs UV, IR and visible radiation types. In molecules, other than the electronic transitions, vibration and rotational transitions are also possible. In atoms, these absorption spectra are seen as a result of electronic transitions. When the transmitted radiation is recorded, the spectrum consists of a number of very narrow absorption lines. Absorbed frequency differs for different atoms. When a beam of polychromatic UV or visible radiation passes through atoms in the gas phase, only some of the frequencies are absorbed by the atoms. Absorption spectra can be of two types as atomic absorption spectra and molecular absorption spectra. Other than the wave length absorbance can also be plotted against frequency or wave number. Therefore, this can be used to identify the chemical species.Īn absorption spectrum is a plot of absorbance versus wavelength. Each element emits specific radiation depending on the energy gap between the ground state and the excited state. This emitted radiation is plotted as a function of frequency or wavelength, and it is then called an emission spectra. This excited state species is unstable therefore, trying to emit energy and come back to the ground state. After applying external energy, some of the molecules will undergo a transition to a higher energy state called the excited state. Before applying energy, the molecules in the sample are in a lower energy state, which we call the ground state. First, the sample is stimulated by applying energy in the form of heat, electrical energy, light, particles, or a chemical reaction. When a sample is given, we can obtain information about the sample depending on its interaction with the radiation. There are different spectrophotometers to measure different types of electromagnetic radiations such as IR, UV, visible, X-ray, microwave, radio frequency, etc. These energies are studied in spectroscopy. Molecules or atoms can absorb energy or release energy. The interaction of radiation and matter is the subject of the science called spectroscopy. Light and other forms of electromagnetic radiations are very useful, and widely used in analytical chemistry. Emission vs Absorption Spectra | Absorption Spectrum vs Emission Spectrum
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
